Introduction to Atomic Structure
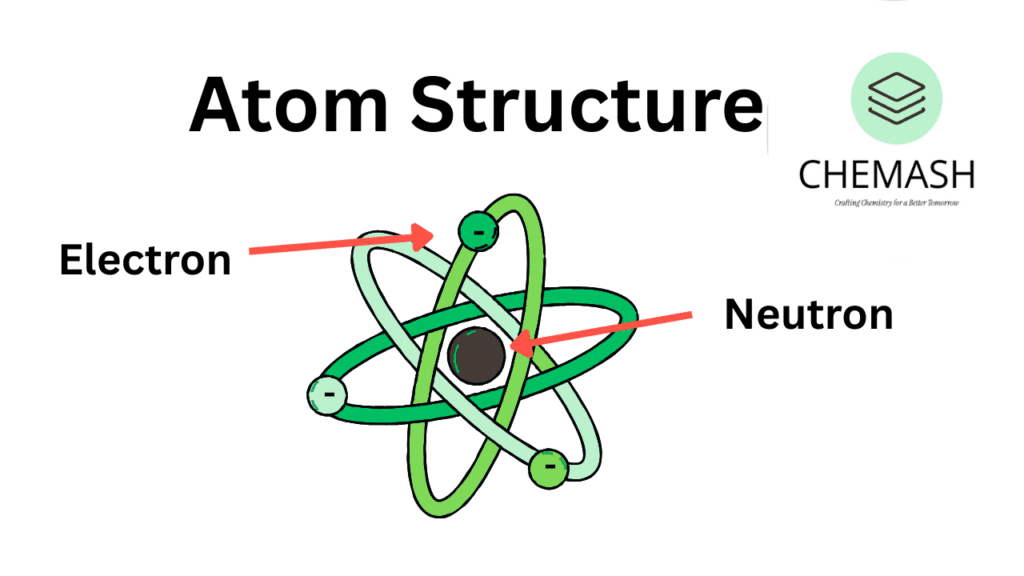
The atomic structure is the foundation of all matter. Atoms are the smallest units that retain the identity of an element. They consist of protons, neutrons, and electrons. Understanding atomic structure helps explain periodic trends, bonding, spectroscopy, and chemical reactivity.
Basic Composition of an Atom
- Protons: Positively charged particles in the nucleus; define atomic number.
- Neutrons: Neutral particles in the nucleus; contribute to atomic mass.
- Electrons: Negatively charged particles in shells around the nucleus.
Modern Understanding
From Dalton’s indivisible sphere to quantum mechanics, our knowledge of atomic structure has evolved. The Quantum Mechanical Model describes electrons as existing in orbitals (regions of probability) instead of fixed orbits.
Figure: Simplified Model of an Atom
Historical Milestones
- Dalton (1803): Atoms are indivisible, indestructible.
- J.J. Thomson (1897): Discovered electron, proposed “plum pudding” model.
- Rutherford (1911): Nuclear model with dense positive nucleus.
- Bohr (1913): Quantized electron orbits to explain spectra.
- Quantum Mechanical Model: Schrödinger & Heisenberg introduced orbitals.
Significance of Atomic Structure
- Explains periodic trends (Electronic Configuration)
- Basis of chemical bonding (Chemical Bonding)
- Helps understand Spectroscopy and transitions
- Explains ions, molecular compounds, and reactivity
MCQ
- Who discovered the electron?
- a) Rutherford
- b) J.J. Thomson ✅
- c) Bohr
- d) Dalton
- What is the charge of a neutron?
- a) Positive
- b) Negative
- c) Zero ✅
- d) Variable
- Which particle defines atomic number?
- a) Electron
- b) Proton ✅
- c) Neutron
- d) Nucleus
- Who proposed electron orbits?
- a) Dalton
- b) Bohr ✅
- c) Thomson
- d) Schrödinger
- Where are protons located?
- a) Shells
- b) Orbitals
- c) Nucleus ✅
- d) Clouds
FAQs on Atomic Structure
1. What is an atom?
An atom is the smallest unit of matter that retains the identity of an element.
2. How many types of subatomic particles are there?
Three: protons, neutrons, and electrons.
3. Who gave the nuclear model of the atom?
Ernest Rutherford in 1911 through the gold foil experiment.
4. What is the latest atomic model?
The Quantum Mechanical Model, which explains electrons in orbitals as regions of probability.
5. Why is atomic structure important in chemistry?
It explains chemical bonding, reactivity, periodic trends, and spectroscopy.
Related reading: | Encyclpedia Britannica on Atomic Theory
