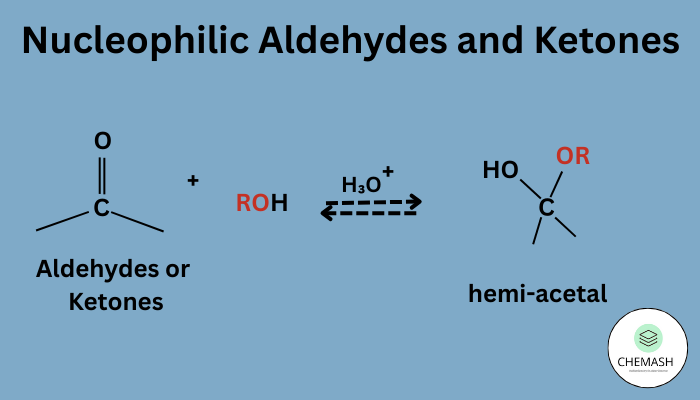
Introduction to Aldehydes and Ketones
Aldehydes and ketones are organic compounds belonging to the carbonyl family, characterized by the highly reactive carbonyl group (C=O). This group influences both reactivity and physical behavior, making aldehydes and ketones essential in organic chemistry, biological systems, and industries.
The Carbonyl Functional Group
The carbonyl group (C=O) consists of a carbon atom double bonded to oxygen. It is:
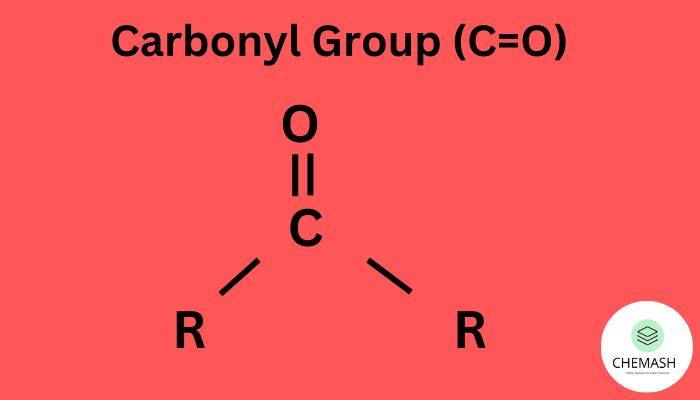
- Planar in geometry with bond angles ≈ 120°
- Polar due to electronegativity difference between C and O
- A key site for nucleophilic and electrophilic reactions
What are Aldehydes?
Aldehydes contain the carbonyl group at the end of a carbon chain, bonded to at least one hydrogen atom.
General Formula: R–CHO
Examples: Formaldehyde (HCHO), Acetaldehyde (CH₃CHO)
They occur in essential oils, flavors, perfumes, and metabolic intermediates like glucose.
What are Ketones?
Ketones feature the carbonyl group bonded to two carbon atoms within the chain.
General Formula: R–CO–R′
Examples: Acetone (CH₃COCH₃), Butanone (CH₃COC₂H₅)
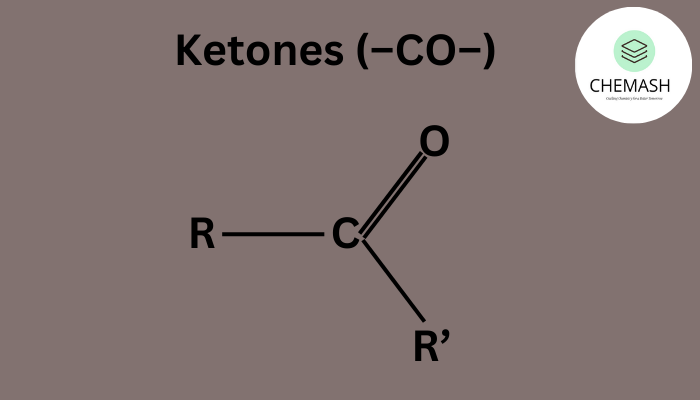
Ketones are used as solvents, in fragrances, and as intermediates in metabolism and industries.
Reactivity & Applications
- Undergo nucleophilic addition reactions due to polar carbonyl group
- Intermediates in synthesis of alcohols, acids, and other compounds
- Important in pharmaceuticals, polymers, and dyes
- Found in natural biomolecules like sugars (glucose & fructose)
Summary
- Aldehydes: R–CHO → terminal carbonyl group
- Ketones: R–CO–R′ → internal carbonyl group
- Contain polar
C=Ogroup affecting physical & chemical properties - Industrial + biological importance
MCQ Quiz – Aldehydes & Ketones
- The functional group in aldehydes is:
a) –COOH
b) –CHO ✅
c) –OH
d) –COOR - Which of the following is a ketone?
a) Formaldehyde
b) Acetone ✅
c) Methanol
d) Ethanal - Aldehydes and ketones mainly undergo:
a) Substitution
b) Nucleophilic addition ✅
c) Elimination
d) Polymerization
True/False
- Carbonyl group is polar. ✅ (True)
- Ketones always have a hydrogen attached to the carbonyl carbon. ❌ (False)
- Aldehydes and ketones are important in industrial chemistry. ✅ (True)
Internal Links:

Pingback: ketones-introduction
Pingback: Chemical Reactions of Aldehydes | Organic Chemistry -