Ionic Equilibrium – Complete Exam Oriented Notes (JEE | NEET | Boards 2026)
CHEMASH presents highly structured, problem-solving focused Ionic Equilibrium notes designed specifically for JEE Main, JEE Advanced, NEET & Class Boards 2026. This chapter is one of the most scoring yet conceptually sensitive areas in Physical Chemistry.
Table of Contents
- Introduction
- Acids, Bases & Their Strength
- pH Scale & Calculations
- Salt Hydrolysis
- Buffer Solutions
- Solubility Product (Ksp)
- JEE & NEET Exam Strategy
- Why CHEMASH?
1️⃣ Introduction
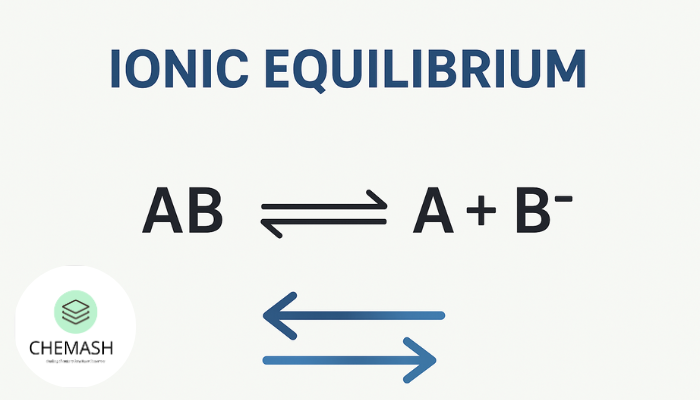
Ionic equilibrium deals with the equilibrium established between ions and unionized molecules in electrolyte solutions. Unlike chemical equilibrium, it focuses on ionization reactions occurring in aqueous media.
- Weak electrolytes establish equilibrium
- Strong electrolytes are almost fully ionized
- Le Chatelier’s principle applies
2️⃣ Acids, Bases & Their Strength
Arrhenius Concept
Acids produce H⁺ ions and bases produce OH⁻ ions in aqueous solution.
Bronsted–Lowry Concept
Acid → Proton donor Base → Proton acceptor
Strength of Acids & Bases
- Ka – Acid dissociation constant
- Kb – Base dissociation constant
- Stronger acid → Higher Ka value
JEE Tip: Logarithmic comparison of Ka values is common.
3️⃣ pH Scale & Calculations
pH is a measure of acidity or basicity of a solution.
pH = −log[H⁺]
pOH = −log[OH⁻]
pH + pOH = 14 (at 25°C)
Numerical Focus:
- Weak acid pH using approximation
- Strong acid/base dilution problems
- Common ion effect
4️⃣ Salt Hydrolysis
Salt hydrolysis depends on the nature of acid and base forming the salt.
| Salt Type | Nature |
|---|---|
| Strong Acid + Strong Base | Neutral |
| Strong Acid + Weak Base | Acidic |
| Weak Acid + Strong Base | Basic |
NEET Favorite: Predicting pH without calculation.
5️⃣ Buffer Solutions
Buffers resist change in pH upon addition of small amounts of acid or base.
Henderson–Hasselbalch Equation
pH = pKa + log (Salt / Acid)
- Acidic buffers → Weak acid + its salt
- Basic buffers → Weak base + its salt
JEE Advanced Focus: Buffer capacity & mixed buffers.
6️⃣ Solubility Product (Ksp)
Ksp represents the extent of solubility of sparingly soluble salts.
For AgCl:
Ksp = [Ag⁺][Cl⁻]
Key Applications:
- Selective precipitation
- Common ion effect
- Solubility calculation
JEE & NEET: One guaranteed numerical.
JEE | NEET | Boards 2026 Strategy
- Memorize log values & formulas
- Practice approximation-based numericals
- Focus on conceptual MCQs
- Revise PYQs (last 15 years)
Frequently Asked Questions
Q1. Is Ionic Equilibrium important for JEE and NEET 2026?
Yes, one of the most important chapters in Physical Chemistry. Every year, 2–3 questions are asked in JEE Main and at least 1–2 direct questions appear in NEET, mainly from pH, buffers, Ksp and salt hydrolysis.
Q2. Which topics of Ionic Equilibrium are most scoring?
The most scoring areas include:
- pH and pOH calculations
- Ka, Kb and relation with pKa
- Buffer solutions (Henderson equation)
- Solubility product (Ksp)
- Common ion effect
Q3. Is Ionic Equilibrium difficult for beginners?
Ionic Equilibrium becomes easy once concepts and approximations are clear. With regular problem-solving practice, students can master this chapter. At CHEMASH, the focus is on simplifying numericals step-by-step.
Q4. How much time should I give to Ionic Equilibrium?
Ideally, students should spend 10–12 days covering theory, numericals, and PYQs thoroughly. Daily numerical practice is essential for confidence.
Q5. Are formulas important in Ionic Equilibrium?
Yes.is a formula-driven chapter. Memorizing formulas along with understanding their derivation helps in faster problem-solving during exams.
Q6. Is Ionic Equilibrium required for Class 11 Boards?
Absolutely. Ionic Equilibrium is a core Class 11 Physical Chemistry chapter and carries significant weight in board examinations, especially theory-based and numerical questions.
Q7. How does CHEMASH help in mastering Ionic Equilibrium?
CHEMASH provides problem-solving based online tuitions with exam-focused notes, numericals, PYQs, and concept clarity for JEE, NEET & Boards across all subjects.
Why Study with CHEMASH?
CHEMASH is a problem-solving focused online tuition platform offering:
- Concept clarity + numericals
- JEE, NEET & Boards preparation
- All Subjects: Chemistry, Physics, Maths & Biology
- Exam-oriented approach
CHEMASH = Learn Smart. Solve Better. Rank Higher.

Pingback: Buffer Solutions - CHEMASH