Markovnikov’s Rule
Markovnikov’s Rule is an important rule in organic chemistry that predicts the orientation of addition reactions of unsymmetrical alkenes with polar reagents such as HX (HCl, HBr, HI).
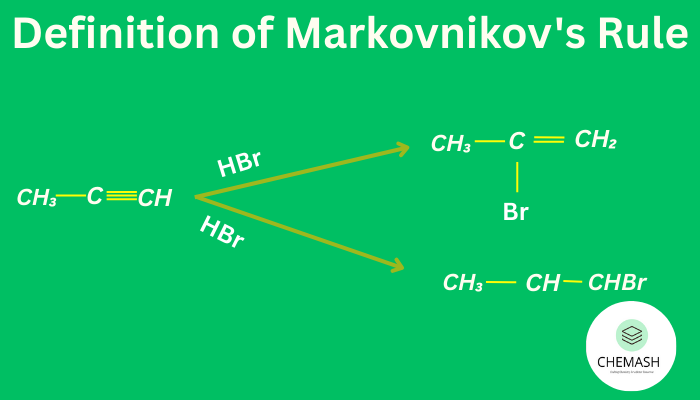
Definition of Markovnikov’s Rule
“In the addition of an unsymmetrical reagent to an unsymmetrical alkene, the hydrogen atom attaches itself to the carbon atom already carrying more hydrogen atoms, while the negative part of the reagent attaches to the carbon atom carrying fewer hydrogen atoms.”
Why Markovnikov’s Rule Works (Mechanism)
The rule is based on the stability of carbocations. During electrophilic addition, the alkene forms a carbocation intermediate. More substituted carbocations are more stable due to:
- +I (inductive) effect
- Hyperconjugation
- Electron-donating alkyl groups
Example of Markovnikov’s Rule
Addition of HBr to propene (CH₃–CH=CH₂):
Hydrogen (H⁺) attaches to the carbon with more hydrogens (terminal carbon), forming a more stable secondary carbocation. Bromide ion (Br⁻) then attaches to the other carbon.
Major product: 2-bromopropane
Anti-Markovnikov Rule (Exception)
In the presence of peroxides (ROOR), addition of HBr to alkenes follows the Anti-Markovnikov rule, where:
- Hydrogen attaches to the carbon with fewer hydrogens
- Bromine attaches to the carbon with more hydrogens
Comparison Table
| Markovnikov Rule | Anti-Markovnikov Rule |
|---|---|
| Based on carbocation stability | Based on free radical mechanism |
| No peroxide required | Peroxide required |
| Applies to HX addition | Mainly applies to HBr only |
MCQs
Q1. The addition of HCl to propene follows Markovnikov’s rule because:
- A. Chloride ion is more reactive
- B. Primary carbocation is more stable
- C. Secondary carbocation is more stable ✅
- D. Reaction is exothermic
Answer: C — Secondary carbocation is more stable.
Frequently Asked Questions
Is Markovnikovs rule always followed?
No. In the presence of peroxides, the rule is not followed and Anti-Markovnikov addition occurs.
Who proposed Markovnikovs rule?
The rule was proposed by Vladimir Markovnikov in 1869.
