Mechanisms of Reactions
In chemistry, a reaction mechanism is a detailed step-by-step description of how a chemical reaction occurs at the molecular level. It explains the breaking and forming of bonds, electron movement, and intermediates. Understanding mechanisms helps chemists predict reaction outcomes.
Key Concepts in Reaction Mechanisms
- Elementary Steps: Small single-step transformations.
- Reaction Intermediates: Temporary species formed during reactions.
- Transition States: High-energy unstable configurations.
- Activation Energy (Ea): Minimum energy needed to start a reaction.
- Rate-Determining Step: Slowest step controlling reaction rate.
Types of Reaction Mechanisms
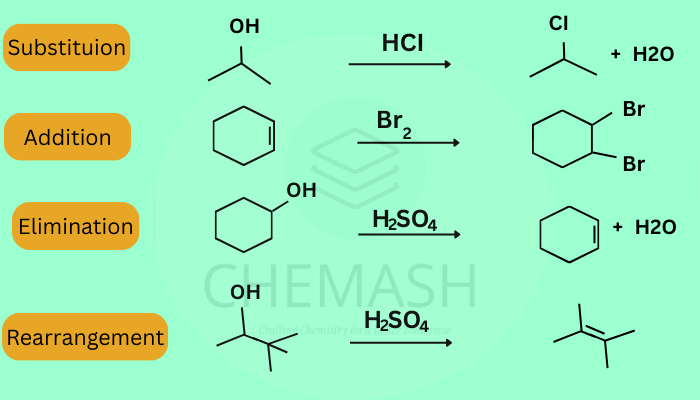
- Substitution Reactions: Replacement of atoms/groups (SN1 & SN2).
- Addition Reactions: Addition to double/triple bonds (alkenes/alkynes).
- Elimination Reactions: Removal of atoms to form multiple bonds.
- Redox Reactions: Electron transfer reactions.
- Free Radical Reactions: Chain reactions involving radicals.
Example: SN2 Mechanism
The SN2 mechanism occurs in one step:
CH3Br + OH− → CH3OH + Br−
Mechanism: OH− attacks from the back → Br− leaves → inversion of configuration.
Reaction Coordinate Diagram
Shows energy profile of reactions:
- Reactants at initial energy.
- Transition state at peak energy.
- Products lower or higher based on exothermic/endothermic nature.
Chain Reaction Example (Free Radical Mechanism)
Chlorination of Methane:
- Initiation: Cl2 → 2Cl• (UV light)
- Propagation: Cl• + CH4 → CH3• + HCl
- CH3• + Cl2 → CH3Cl + Cl•
- Termination: Radicals combine to stop reaction.
Conclusion
Reaction mechanisms explain how reactions occur at the molecular level, crucial for organic synthesis, pharmaceuticals, and materials science.
Quick MCQs
Q1: Which step controls the overall rate of a reaction?
A) Initiation
B) Propagation
C) Rate-determining step ✅
D) Transition state
Q2: SN2 mechanism involves:
A) Two steps
B) One step ✅
C) Free radicals
D) Carbocation
Fill in the Blanks
1. The minimum energy needed to start a reaction is called _______. (Activation Energy)
2. In an SN2 reaction, the nucleophile attacks from the ______ side. (Back)
True/False
1. Free radical reactions involve unpaired electrons. ✅
2. Elimination reactions always form single bonds. ❌
FAQs
Q1: Why is SN2 a one-step mechanism?
Ans: Because bond-breaking and bond-making occur simultaneously.
Q2: What is the role of the transition state?
Ans: It represents the highest energy point before product formation.
