Tests for Carboxylic Acids | कार्बोक्सिलिक अम्ल की जांच
Table of Contents
- Litmus Test
- Sodium Bicarbonate Test
- Reaction with NaOH
- Esterification Test
- Ferric Chloride Test
- IR Spectroscopy
- MCQs
- Fill in the Blanks
- Quiz
- FAQs
Litmus Test | लिटमस परीक्षण
Carboxylic acids turn blue litmus paper red due to their acidic nature.
Sodium Bicarbonate Test | सोडियम बाइकार्बोनेट परीक्षण
Carboxylic acids react with NaHCO₃ to release CO₂ gas, producing effervescence.
Reaction with NaOH | NaOH के साथ प्रतिक्रिया
Carboxylic acids neutralize sodium hydroxide to form sodium salts and water.
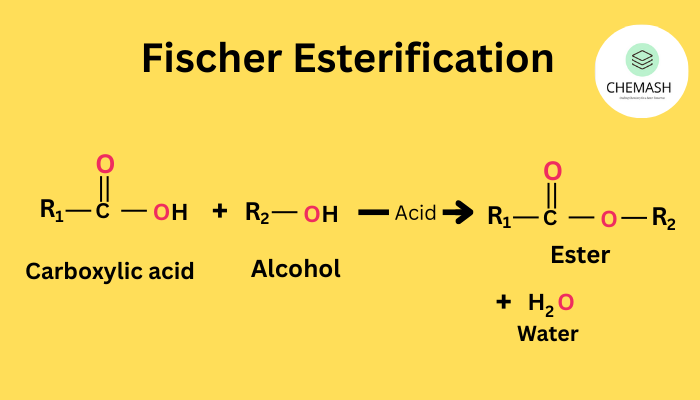
Esterification Test | एस्टरीफिकेशन परीक्षण
Carboxylic acids react with alcohols in presence of conc. H₂SO₄ to form esters, which have fruity smell.
Ferric Chloride Test | फेरिक क्लोराइड परीक्षण
Some carboxylic acids (like aromatic hydroxy acids) give colored complexes with FeCl₃.
IR Spectroscopy | IR स्पेक्ट्रोस्कोपी
Carboxylic acids show a broad absorption band around 2500–3300 cm⁻¹ for O–H stretch and a strong C=O band near 1700 cm⁻¹.
Multiple Choice Questions (MCQs)
1. Which test confirms the presence of carboxylic acids by effervescence?
- A. Litmus Test
- B. Sodium Bicarbonate Test ✅ Correct
- C. Esterification Test
- D. Ferric Chloride Test
2. Carboxylic acids turn blue litmus:
- A. Blue ✅ Correct: Acids turn blue litmus red
- B. Red
- C. Green
- D. Yellow
Fill in the Blanks
1. Carboxylic acids react with ______ to produce CO₂ gas. (NaHCO₃)
2. The fruity smell test of carboxylic acids is called ______. (Esterification)
3. In IR spectroscopy, –COOH group shows a broad band around ______. (2500–3300 cm⁻¹)
Quick Quiz
- Explain why effervescence occurs in sodium bicarbonate test for carboxylic acids.
- Why do esters formed from carboxylic acids have fruity odor?
- How does IR spectroscopy confirm presence of –COOH group?
FAQs (English + Hindi)
Q1. How to quickly test a carboxylic acid in lab?
A: Use sodium bicarbonate test. Effervescence due to CO₂ confirms presence.
कार्बोक्सिलिक अम्ल की पहचान प्रयोगशाला में सोडियम बाइकार्बोनेट से की जाती है। गैस का निकलना पुष्टि करता है।
Q2. Why do carboxylic acids show fruity smell in ester test?
A: Esters formed have characteristic fruity smell.
एस्टर बनने पर फल जैसी गंध आती है।
Q3. Which spectroscopy is best for identifying –COOH group?
A: IR spectroscopy due to O–H and C=O stretching bands.
IR स्पेक्ट्रोस्कोपी सबसे उपयुक्त है।
