Types of Salts
Salts are chemical compounds that arise when an acid reacts with a base. They consist of cations (from bases) and anions (from acids). Because salts vary in composition, origin, and chemical behavior, scientists classify them into several types. By learning these types, students can better predict solubility, reactivity, and real-world applications.
1. Normal Salts
Normal salts form when all hydrogen ions (H⁺) in an acid get replaced by metal or ammonium ions from a base.
Example: NaCl (Sodium chloride), K₂SO₄ (Potassium sulfate)
Reaction: HCl + NaOH → NaCl + H₂O
2. Acid Salts
When only some hydrogen ions of a polybasic acid are replaced, the product is an acid salt. These salts still contain replaceable hydrogen, so they behave like acids.
Example: NaHSO₄ (Sodium bisulfate), NaHCO₃ (Sodium bicarbonate)
Reaction: H₂SO₄ + NaOH → NaHSO₄ + H₂O
3. Basic Salts
Basic salts appear when an acid cannot fully neutralize a base. Consequently, hydroxide ions (OH⁻) remain, giving the salt a basic character.
Example: Mg(OH)Cl (Magnesium hydroxide chloride), Zn(OH)Cl
Reaction: Zn(OH)₂ + HCl → Zn(OH)Cl + H₂O
4. Double Salts
Two different salts can crystallize together, forming double salts. In water, they dissociate into multiple ions.
Example: Mohr’s salt [FeSO₄·(NH₄)₂SO₄·6H₂O], Potash alum [KAl(SO₄)₂·12H₂O]
5. Mixed Salts
Mixed salts include more than one acidic or basic radical. They come from neutralizing more than one acid or base together.
Example: NaKCO₃ (Sodium potassium carbonate), CaOCl₂ (Calcium oxychloride)
6. Complex Salts
Complex salts result from combining a simple salt with a complex ion. Unlike normal salts, they do not fully dissociate into simple ions in water.
Example: K₄[Fe(CN)₆] (Potassium ferrocyanide), [Cu(NH₃)₄]SO₄
7. Hydrated Salts
Some salts hold water molecules in their crystal structure, which scientists call water of crystallization.
Example: CuSO₄·5H₂O (Blue vitriol), MgSO₄·7H₂O (Epsom salt)
8. Anhydrous Salts
When salts lose their water of crystallization, usually through heating, they become anhydrous salts. Because of this, they often absorb water and act as drying agents.
Example: Anhydrous CuSO₄ (white), used as a test for water.
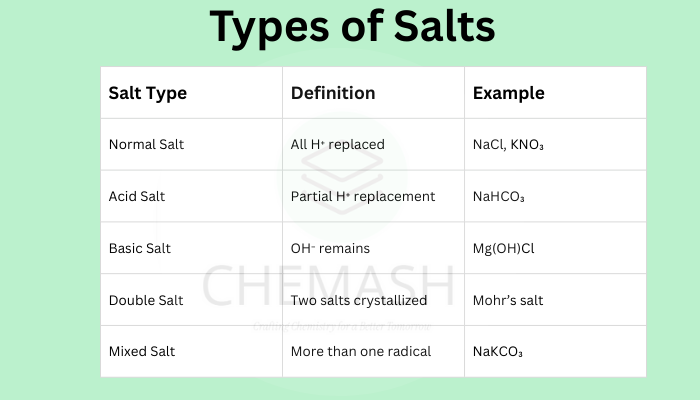
Summary Table
| Salt Type | Definition | Example |
|---|---|---|
| Normal Salt | All H⁺ replaced | NaCl, KNO₃ |
| Acid Salt | Partial H⁺ replacement | NaHCO₃ |
| Basic Salt | OH⁻ remains | Mg(OH)Cl |
| Double Salt | Two salts crystallized | Mohr’s salt |
| Mixed Salt | More than one radical | NaKCO₃ |
Quiz: Types of Salts
Q1: Which salt is an acid salt?
A) NaCl
B) NaHCO₃
C) K₂SO₄
D) NH₄Cl
Answer: B) NaHCO₃
Explanation: Sodium bicarbonate forms through partial neutralization and keeps replaceable hydrogen.
Q2: Which salt results from incomplete neutralization of a base?
A) Basic salt
B) Acid salt
C) Normal salt
D) Hydrated salt
Answer: A) Basic salt
Explanation: Basic salts retain OH⁻ ions, showing incomplete neutralization.
Learn more about Salts on Britannica.

Pingback: Neutralization Reactions - CHEMASH