Homogeneous and Heterogeneous Equilibria
Homogeneous and Heterogeneous Equilibria are two fundamental types of chemical equilibrium that describe how reactions reach balance depending on the phases of reactants and products. In homogeneous equilibrium, all substances are in the same phase (such as gases or aqueous solutions), while in heterogeneous equilibrium, reactants and products exist in different phases. Understanding these equilibria is crucial in predicting reaction behavior in laboratory and industrial processes.
Chemical equilibrium is a fundamental concept in chemistry where the rate of forward and backward reactions becomes equal. It is mainly classified into two types: homogeneous equilibrium and heterogeneous equilibrium. Understanding these is crucial for chemical industries and academic learning.
Table of Contents
- Homogeneous Equilibrium
- Heterogeneous Equilibrium
- Key Differences
- Equilibrium Constant Expressions
- Quiz
- MCQs
- FAQs
1. Homogeneous Equilibrium
In a homogeneous equilibrium, all reactants and products exist in the same phase (gas or aqueous).
- Involves reactions where all species are gases or in solution.
- Equilibrium constant includes all species in the same phase.
Example: Haber process: N2(g) + 3H2(g) ⇌ 2NH3(g)
2. Heterogeneous Equilibrium
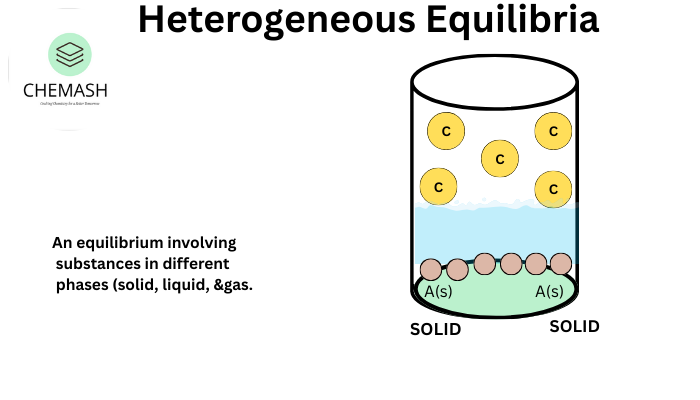
In a heterogeneous equilibrium, reactants and products are in different phases.
- Pure solids and liquids are excluded from the equilibrium constant expression.
- Only gases and aqueous species are included.
Example: CaCO3(s) ⇌ CaO(s) + CO2(g)
Key Differences
| Aspect | Homogeneous | Heterogeneous |
|---|---|---|
| Phases | Same phase | Different phases |
| K Expression | All species | Only gases/aqueous |
| Example | N2 + 3H2 ⇌ 2NH3 | CaCO3(s) ⇌ CaO(s) + CO2(g) |
Equilibrium Constant Expressions
Homogeneous: Kc = [NH3]^2 / ([N2] [H2]^3)
Heterogeneous: Kc = [CO2]
Quiz
- Define homogeneous equilibrium with an example.
- Why are solids excluded in heterogeneous equilibrium?
- Write Kc for
CaCO3(s) ⇌ CaO(s) + CO2(g).
MCQs
- In heterogeneous equilibrium, which is excluded from K?
(a) Gases (b) Aqueous (c) Solids (✔) (d) All - Which is homogeneous?
(a) CaCO3(s) ⇌ CaO(s)+CO2(g) (b) N2+3H2 ⇌ 2NH3 ✔ (c) H2O(l) ⇌ H2+O2
FAQs
Q1: What is homogeneous equilibrium?
A: When all reactants/products are in the same phase.
Q2: Why exclude solids/liquids from K?
A: Their concentration remains constant.
Related: Chemical Equilibrium – Wikipedia
