Chemical Kinetics: Rate and Mechanism of Chemical Reactions
Chemical Kinetics is an important branch of Physical Chemistry that deals with the study of the rate of chemical reactions and the mechanism by which reactions occur.
Unlike thermodynamics, which predicts whether a reaction is feasible, chemical kinetics explains how fast a reaction occurs and how it proceeds.
This chapter is extremely important for Class 11, Class 12, NEET, JEE, and other competitive examinations.
Table of Contents
- Reaction Rate
- Factors Affecting Reaction Rate
- Rate Law and Order of Reaction
- Reaction Mechanism
- Activation Energy and Arrhenius Equation
- Importance of Chemical Kinetics
- MCQs
- FAQs
Reaction Rate
The reaction rate is defined as the speed at which reactants are converted into products. It is usually expressed as the change in concentration per unit time.
Mathematical Expression:
Rate = −Δ[Reactant]/Δt = Δ[Product]/Δt
The negative sign indicates the decrease in reactant concentration with time.
Factors Affecting Reaction Rate
- Concentration: Higher concentration increases collision frequency.
- Temperature: Increases kinetic energy, resulting in faster reactions.
- Catalyst: Lowers activation energy and increases reaction speed.
- Surface Area: Larger surface area of solids increases reaction rate.
- Nature of Reactants: Ionic reactions are generally faster than covalent reactions.
Rate Law and Order of Reaction
The rate law expresses the relationship between reaction rate and reactant concentrations.
General Rate Law:
Rate = k[A]m[B]n
Where:
- k = rate constant (temperature dependent)
- m, n = order of reaction with respect to reactants
- Overall order = m + n
Reaction Mechanism
A reaction mechanism is a step-by-step sequence of elementary reactions that explains how reactants are converted into products.
- The slowest step is called the rate-determining step.
- Intermediates may form temporarily.
- Intermediates do not appear in the overall balanced equation.
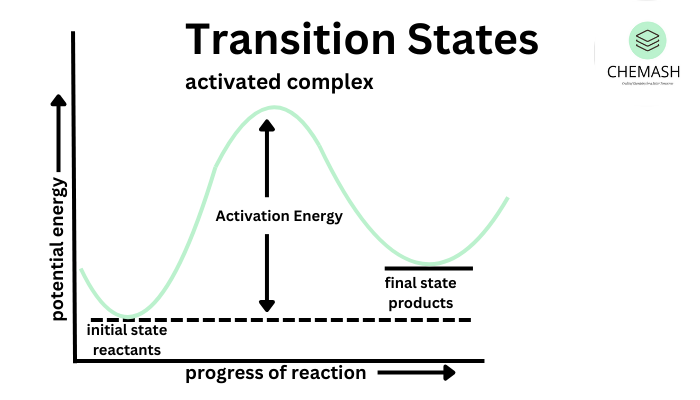
Activation Energy and Arrhenius Equation
Activation energy (Ea) is the minimum energy required for a reaction to occur.
The effect of temperature on reaction rate is explained by the Arrhenius equation.
Arrhenius Equation:
k = A e−Ea/RT
Where:
- k = rate constant
- A = frequency factor
- Ea = activation energy
- R = gas constant
- T = temperature (Kelvin)
As temperature increases, the rate constant increases, making reactions faster.
Importance of Chemical Kinetics
- Industrial Chemistry: Optimization of manufacturing processes
- Biochemistry: Understanding enzyme action and drug design
- Environmental Science: Studying pollutant degradation
- Safety Engineering: Predicting hazardous and explosive reactions
Exam-Oriented MCQs
Q1. Which branch of chemistry deals with reaction rate?
A) Chemical Kinetics B) Thermodynamics C) Electrochemistry D) Nuclear Chemistry
Q2. The slowest step in a reaction mechanism is called:
A) Fast step B) Rate-determining step C) Initiation step D) Termination step
Frequently Asked Questions (FAQs)
Q. What is chemical kinetics?
Answer: Chemical kinetics is the study of reaction rates and mechanisms.
Q. How is chemical kinetics different from thermodynamics?
Answer: Thermodynamics predicts feasibility, while kinetics predicts reaction speed.
- Rate of Reaction
- Collision Theory
- Integrated Rate Laws
- Chemistry LibreTexts
- Khan Academy – Chemistry
© 2025 CHEMASH | Physical Chemistry

Pingback: Physical Chemistry क्या है? - CHEMASH