Chromatography Techniques – Principle, Types, Applications .
Chromatography techniques are among the most important separation methods in analytical chemistry. They allow scientists to separate, identify, purify, and quantify components of a mixture. Because of their accuracy and versatility, chromatographic methods are widely used in organic chemistry, biochemistry, pharmaceuticals, environmental science, and forensic analysis.
Within the CHEMASH Analytical Chemistry SILO, chromatography works in close coordination with Mass Spectrometry (MS), NMR Spectroscopy, and Infrared Spectroscopy. Therefore, students and researchers obtain complete molecular identification.
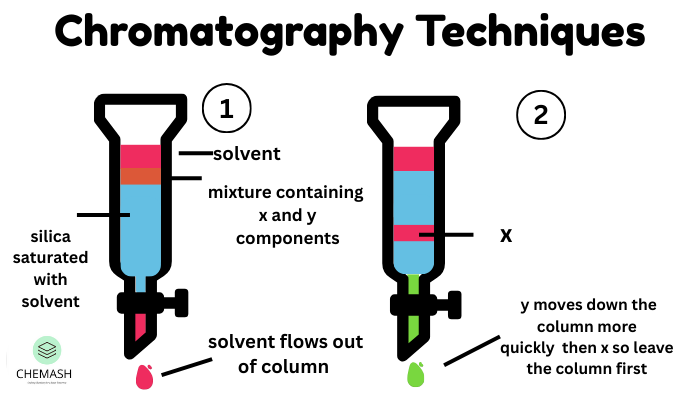
Table of Contents
- Introduction to Chromatography
- Principle of Chromatography
- Classification of Chromatography
- Adsorption Chromatography
- Partition Chromatography
- Paper Chromatography
- Thin Layer Chromatography (TLC)
- Column Chromatography
- Gas Chromatography (GC)
- High Performance Liquid Chromatography (HPLC)
- Other Chromatographic Techniques
- Applications of Chromatography
- Advantages
- Limitations
- Frequently Asked Questions
Introduction to Chromatography
Chromatography is a physical separation technique based on the differential distribution of components between two phases: a stationary phase and a mobile phase. As the mobile phase moves, different components travel at different rates. Consequently, separation occurs.
Russian scientist Mikhail Tswett introduced chromatography in 1903 while separating plant pigments. Since then, chromatographic techniques have evolved significantly and now form the backbone of modern analytical laboratories.
Principle of Chromatography
The fundamental principle of chromatography depends on differences in adsorption, partition, ion exchange, molecular size, or affinity. Because components interact differently with stationary and mobile phases, they separate over time.
In simple terms:
Component with stronger interaction → slower movement
Component with weaker interaction → faster movement
Therefore, chromatographic separation remains highly selective and reproducible.
Classification of Chromatography Techniques
Chromatography techniques can be classified based on the nature of stationary and mobile phases as well as the separation mechanism.
- Adsorption chromatography
- Partition chromatography
- Ion-exchange chromatography
- Gel permeation chromatography
- Affinity chromatography
Adsorption Chromatography
In adsorption chromatography, solid adsorbents such as silica gel or alumina act as stationary phases. The separation depends on the degree of adsorption of components.
Examples include:
- Column chromatography
- Thin layer chromatography
Partition Chromatography
Partition chromatography involves a liquid stationary phase immobilized on a solid support. Components distribute themselves between two immiscible liquids.
Paper chromatography and gas chromatography belong to this category.
Paper Chromatography
uses cellulose paper as the stationary phase. It is primarily used for separating amino acids, sugars, and inks.
The Rf value plays a crucial role:
Rf = Distance travelled by solute / Distance travelled by solvent
Thin Layer Chromatography (TLC)
Thin layer chromatography employs a thin layer of adsorbent coated on glass or aluminum plates. Because TLC offers rapid separation and high sensitivity, it is widely used in laboratories.
Applications of TLC include:
- Monitoring reaction progress
- Checking purity of compounds
- Identifying components using Rf values
Column Chromatography
Column chromatography separates compounds using a packed column containing adsorbent. As solvent flows downward, components elute at different times.
This technique is extensively used for purification of organic compounds.
Gas Chromatography (GC)
Gas chromatography uses an inert gas as the mobile phase. It is ideal for volatile and thermally stable compounds.
GC often couples with mass spectrometry, forming GC–MS for highly accurate analysis.
High Performance Liquid Chromatography (HPLC)
HPLC uses high-pressure pumps to pass liquid mobile phase through a packed column. It provides excellent resolution and fast analysis.
HPLC finds extensive use in pharmaceutical quality control and biochemical analysis.
Other Chromatographic Techniques
- Ion exchange chromatography
- Gel permeation chromatography
- Affinity chromatography
Applications of Chromatography
Chromatography plays a vital role across scientific disciplines.
- Drug analysis and quality control
- Environmental pollutant detection
- Biomolecule separation
- Forensic investigations
- Food and beverage analysis
Authoritative references: Britannica, ScienceDirect.
Advantages of Chromatography
- High separation efficiency
- Wide applicability
- High sensitivity
- Accurate qualitative and quantitative analysis
Limitations of Chromatography
Although chromatography is powerful, it has limitations. Some techniques require expensive instrumentation. Moreover, sample preparation can be time-consuming.
Frequently Asked Questions
What is the main principle of chromatography?
Chromatography separates components based on differential distribution between stationary and mobile phases.
Which chromatography is best for purification?
Column chromatography and HPLC are widely used for purification.
What does Rf value indicate?
Rf value indicates the relative movement of a compound in chromatography.
© CHEMASH – Advanced Chemistry Learning Platform
